
The empirical model thus created was statistically relevant. Minitab19 statistical software was utilized for experimentation design and analysis. 
Focused beam reflectance measurements were used to obtain crystal morphology data. This paper presents the implementation of a DoE driven QbD approach for the anti-solvent crystallization of Dexlansoprazole API. Many problems in downstream processes can be attributed to poor particle characteristics established during the crystallization step. In recent years, crystallization of active pharmaceutical ingredients (APIs) has been explored to generate crystals with desirable physical characteristics.  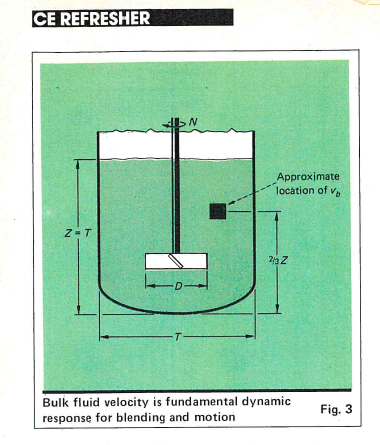
Use of statistical design of experiments (DoE) is widely accepted to be a more efficient experimental strategy for gaining the required process understanding than the traditional one-variable-at-a-time approach. Developing an understanding of critical process parameters and their impact on product quality attributes is crucial for implementing quality-by-design (QbD) in a pharmaceutical crystallization process.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
